Microsoft's Majorana 1 quantum computing chipMicrosoftAfter decades confined largely to research labs, Quantum computing may be closer to its breakout moment than many on...
AUSTIN, Texas — The University of Texas System Board of Regents today approved the creation of a new School of...
Apple's iOS 27 update will prioritize cleaning up the operating system's internals, with engineers making changes that could result in...
In October 2025, there was a groundbreaking scientific breakthrough that might have passed you by. Google...
No matter what part of the internet I've been on, I've always seen folks praising Linux for doing what Windows...
Investors interested in Computer and Technology stocks should always be looking to find the best-performing companies in the group. Advanced...
QUERSUS is a Polish brand founded in 2016 by Miłosz Leśniak and Andrzej Wnękowicz. It focuses on the production of...
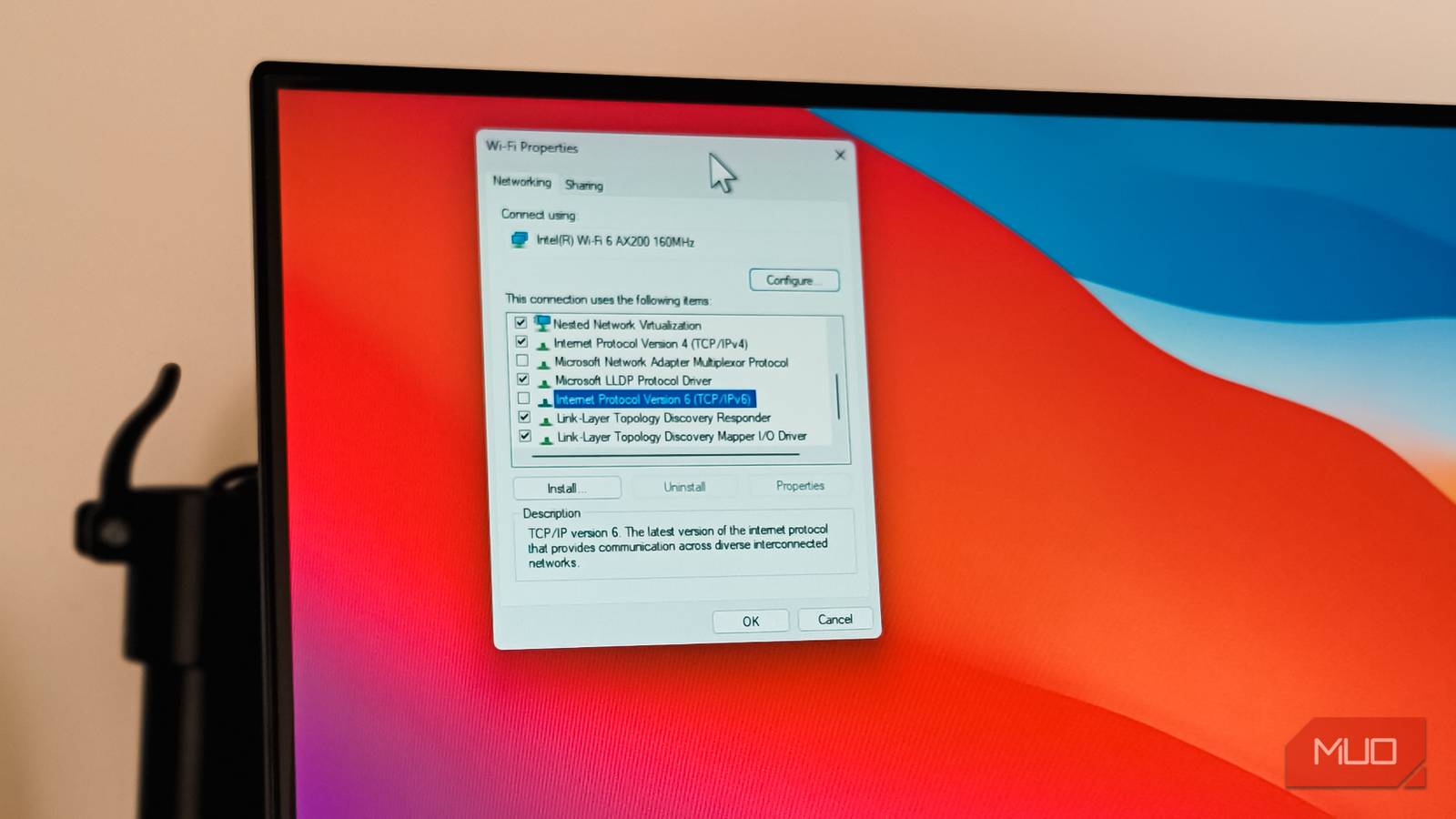
If you're on a slow internet connection, you'd expect your YouTube videos to buffer. But what if videos start buffering...
Summary Fedora Pocketblue Remix brings an atomic Fedora (OCI/OSTree/Bootc) to phones/tablets. Still a WIP: only confirmed on Xiaomi Pad 5,...